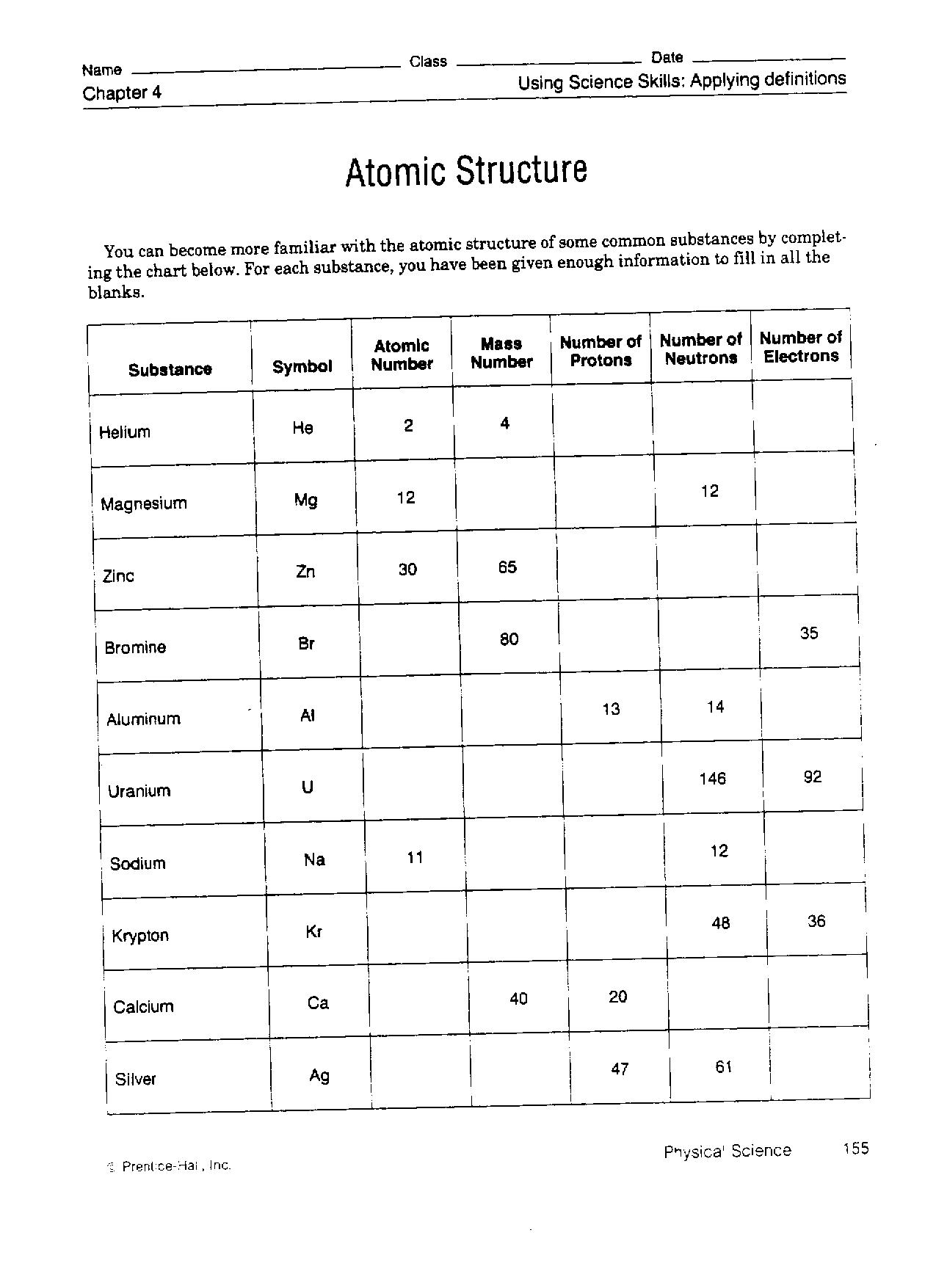 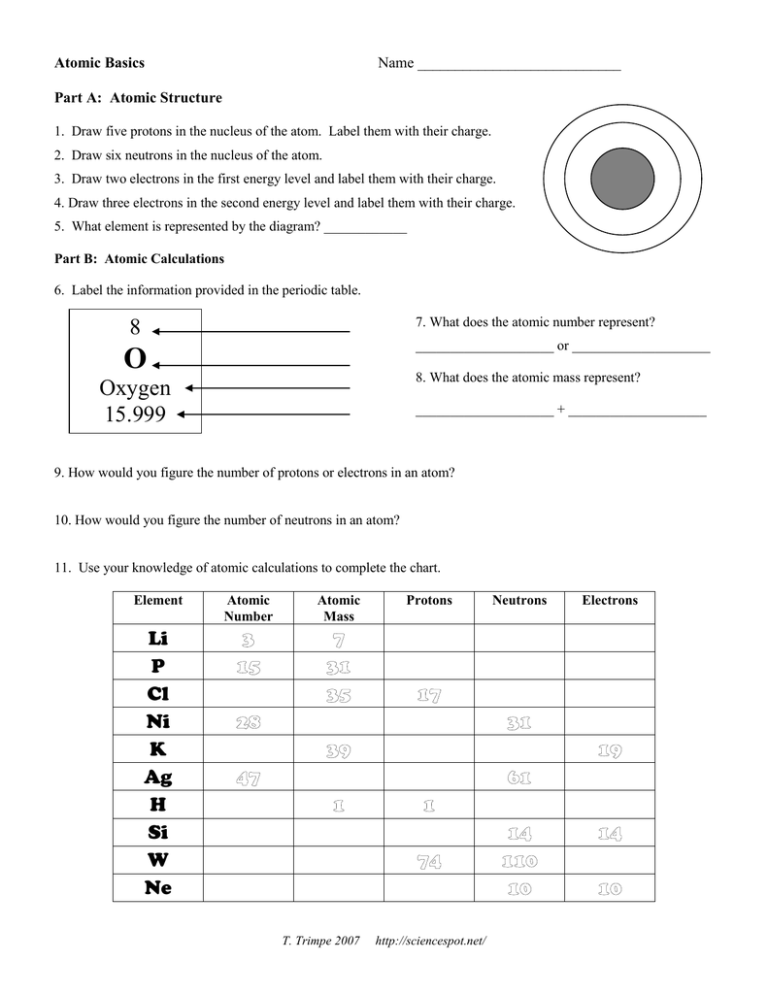
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. These established that atoms can be further divided into sub- atomic particles, i.e., electr ons, pr otons and neutr ons a concept very different from that of Dalton. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. These concepts form the basis of chemistry. Department of Energys (DOE) Brookhaven National Laboratory have determined the atomic-level structure of a zinc-transporter. Atoms combine in whole-number ratios to form compounds. 19 hours ago &0183 &32 Reviewed by Danielle Ellis, B.Sc. Atoms of the same element are the same atoms of different elements are different. The nucleus is small and dense compared with the electrons, which are the lightest charged particles in nature. The numbers of subatomic particles in an atom can be. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. It consists of three parts: All matter is composed of atoms. The rest consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. GCSE AQA Trilogy Atomic structure - AQA Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
